The second law of thermodynamics (the entropy law or law of entropy) was formulated in the middle of the last century by Clausius and Thomson following Carnot's earlier observation that, like the fall or flow of a stream that turns a mill wheel, it is the "fall" or flow of heat from higher to lower temperatures that motivates a steam engine. The key insight was that the world is inherently active, and that whenever an energy distribution is out of equilibrium a potential or thermodynamic "force" (the gradient of a potential) exists that the world acts spontaneously to dissipate or minimize. All real-world change or dynamics is seen to follow, or be motivated, by this law. So whereas the first law expresses that which remains the same, or is time-symmetric, in all real-world processes the second law expresses that which changes and motivates the change, the fundamental time-asymmetry, in all real-world process. Clausius coined the term "entropy" to refer to the dissipated potential and the second law, in its most general form, states that the world acts spontaneously to minimize potentials (or equivalently maximize entropy), and with this, active end-directedness or time-asymmetry was, for the first time, given a universal physical basis. The balance equation of the second law, expressed as S > 0, says that in all natural processes the entropy of the world always increases, and thus whereas with the first law there is no time, and the past, present, and future are indistinguishable, the second law, with its one-way flow, introduces the basis for telling the difference.
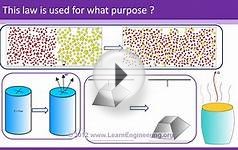
The active nature of the second law is intuitively easy to grasp and empirically demonstrate. If a glass of hot liquid, for example, as shown in Figure 3, is placed in a colder room a potential exists and a flow of heat is spontaneously produced from the cup to the room until it is minimized (or the entropy is maximized) at which point the temperatures are the same and all flows stop.
A glass of liquid at temperature TI is placed in a room at temperatureTII such that . The disequilibrium produces a field potential that results in a flow of energy in the form of heat from the glass to the room so as to drain the potential until it is minimized (the entropy is maximized) at which time thermodynamic equilibrium is reached and all flows stop. refers to the conservation of energy in that the flow from the glass equals the flow of heat into the room. (From Swenson, 1991a. Copyright 1991 Intersystems Publications. Adapted by permission).
Figure 4 shows various other potentials and the flows they would produce. Of important theoretical interest for this paper is the fact that Joule's experiment (Figure 2) while designed to
Further examples of potentials that follow from nonequilibrium distributions of energy. Whenever energy (in whatever form) is out of equilibrium with its surroundings, a potential exists for producing change that, following the second law, is spontaneously minimized.
What does it mean to be in love with someone? How to pick a good cantaloupe? What does pilialoha meaning hawaiian? How to frost tips sponge twists? how to convert wii usb helper to wud What does vapid mean? What is an ovarian cyst? How to make myself cum? What is today's weather forecast? How many free credit reports are you legally entitled to each year?? What is the meaning of the letter s? What does prehistoric mean? What is the meaning of sakura in japanese? What does dementia mean? How to go vegan? How to get rid of the hiccups? Powerball how to play? What does being drunk feel like? How to get healthy hair? How to cook a ribeye? How to cook.steak tips? How to change text size on iphone? What does redeemed mean? What does git mean? What does cousin once removed mean? How to make a sourdough starter? how do uhaul moving helper work What is the meaning of unremarkable in medical term? What does mirandize mean? How to print double sided on mac? Tricks on how to keep outlook 2010 from not responding? How to cook tri tip in the oven? What time does iowa state play today? How to refinance student loans? What does intergate mean? How long does it take to steam broccoli? how to be santa's helper What was the meaning of encanto movie? How to replace tips on pool cues? How to grow breast in 2 days? What time does dairy queen close? What is the true meaning of the name monica? Tips on how to have masturbation be more enjoyable but not cum as fast? What is the meaning of information literacy? See what i'm missing imma need some of that meaning? How to remove sap from car? How to make sorority merch tips? Tips for singers who want emotions? What is an antagonist? how do u define helper function in hashkell What is the meaning of hamlet as a whole? how to make a helper t cell with paper How to make different stick bomb tricks? What does a tick look like in skin? How deep to plant potatoes? Magicians who blundered their tricks? Tips for kids who have problems learning? What is the meaning of medallion? How to raise fico score tricks of a credit hacker? How to get rid of clogged pores? What is shopify? How much does it cost to declaw a cat? How to serve in tennis? What does etf mean? What does the phrase how's tricks mean? What does lush mean? What is the meaning of antecedent? What is the meaning of accountable? How to day trade? What is the full meaning of warda? How to measure square feet? How to catch a smuggler? What is the meaning of mining cryptocurrency? What does unfettered mean? What is the meaning of on pins and needles? What animal does steak come from? How to spoon? How to do your nails with just nail tips? What is the meaning of optician? What is let's go brandon'' mean? What are the five food groups? What does the zodiac sign pisces mean? what does helper mean in rpg game What does motility mean? What is the biblical meaning of barbara? What does dad stand for? What is the spiritual meaning of 111? What is the meaning of yellow tulips? Tips on how to get off of level 125 in candy crush soda? Why don't business allow their employees to take tips? What does the world tarot card mean? How to build chest muscles? Oh how the tables have turned meaning? What is a grifter? How to save instagram photos? What a bust meaning? What is the meaning of emporium? What is the meaning of the name valeria? What does fear stand for?