| Developing a Coherent Approach
A frequent question on the AP Physics B Exam deals with what is commonly referred to as a P-V diagram. An ideal gas undergoes changes of state as a result of work processes, heat processes, or both. The pressure of the gas is then plotted versus volume, and questions are asked about the state of the gas and how much work is done or heat is transferred during the different processes. As common as these questions are on the exam, they are not always a point of emphasis in the various textbooks used for the B-level course. The object of this article is to present a coherent approach to this material based on the ideal gas law and the first law of thermodynamics. Students typically learn about mechanical energy during the first semester of the B-level course. Analysis of the work necessary to accelerate an object or to reposition the object leads to the concepts of kinetic and potential energy. Should only conservative forces act on an object, the sum of the kinetic and potential energies will not change. This sum, the total mechanical energy, is associated with macroscopic motions. However, the objects we use in class to demonstrate mechanical energy (balls, textbooks, weights, springs, etc.) are all complicated systems composed of many molecules or atoms. Even when the center of mass of these objects is stationary, there is considerable energy within the object due to molecular motions and positions. I keep a small demonstration piece consisting of a dozen small spheres interconnected by springs on my front desk. Using this Einstein model of a solid, I can demonstrate kinetic and potential energy at the molecular level. The sum total of the kinetic and potential energy at the molecular level is called the internal energy of the system: U = KE+PE (molecular level). Of course, the springs in the demo piece are representative of an electric interaction between the molecules, and in a solid this interaction is strong enough to keep each molecule close to its equilibrium position. In a liquid, the molecules are still very close to each other, but the interaction is not strong enough to keep the molecules at fixed lattice sites. In a gas, the interaction is relatively weak (more precisely, the interaction energy is small compared to the average kinetic energy of a molecule), and the molecules can readily move throughout the enclosed volume if the density of the gas is not too great. In an ideal gas, the interaction energy between molecules is neglected, that is, there is no internal potential energy. This means that the internal energy of an ideal gas is the sum total of the kinetic energy of the molecules. A standard derivation in most textbooks based on kinetic molecular theory relates the average kinetic energy of a molecule to the temperature: . In this equation, is Boltzmann's constant, , and temperature is expressed... |

What is the First Law of Thermodynamics? - Chemistry Tips

Chapter 15, Example #2 (First Law of Thermodynamics)






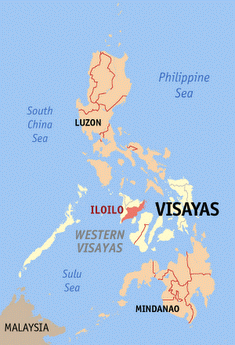 Iloilo is a province of the Philippines located in the Western Visayas region. Iloilo occupies the southeast portion of Panay Island and is bordered by Antique Province to the west and Capiz Province and the Jintotolo Channel to the north. Just off Iloilo's...
Iloilo is a province of the Philippines located in the Western Visayas region. Iloilo occupies the southeast portion of Panay Island and is bordered by Antique Province to the west and Capiz Province and the Jintotolo Channel to the north. Just off Iloilo's...