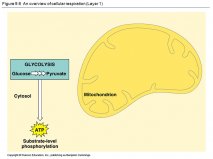
The Second Law of Thermodynamics, formulated in the 19th century, specifies that in a closed system, order must either stay constant or decrease.Dust off your physics knowledge-this is a straightforward application of the second law of thermodynamics, says Lstiburek.In this article, I argue that the second law of thermodynamics is of special importance even within the domain of the humanist, especially in the evolution of culture.Key Topics Covered: Lab 1: Introduction to Heat and Temperature Lab 2: Energy Transfer and Temperature Change Lab 3: Heat Energy Transfer Lab 4: The First Law of Thermodynamics Lab 5: The Ideal Gas Law Lab 6: Heat Engines Appendix A: RealTime Physics Heat and Thermodynamics Experiment Configuration Files For more information visit Among the topics are properties of pure substances, the second law of thermodynamics and entropy, vapor power systems, and principles of energy transfer.However, the second law of thermodynamics requires that heat-driven machines receive energy that raises their temperatures above that of the surroundings-so that the flow of heat from hot to cold can run the machines (SN: 10/7/00, p.Their topics include the first law of thermodynamics, availability, postulatory or Gibbsian thermodynamics, state relationships for real gases and liquids, thermodynamic properties of pure fluids and mixtures, phase equilibrium for a mixture, stability, reaction direction and chemical equilibrium, analyzing availability for reacting systems, and thermodynamics and biological systems.

First Law of Thermodynamics, Lecture 2, Physics IIT JEE ...

Physical Chemistry Lecture: The First Law of Thermodynamics

Physics - Thermodynamics: (1 of 22) First Law of ...
first law of thermodynamics